


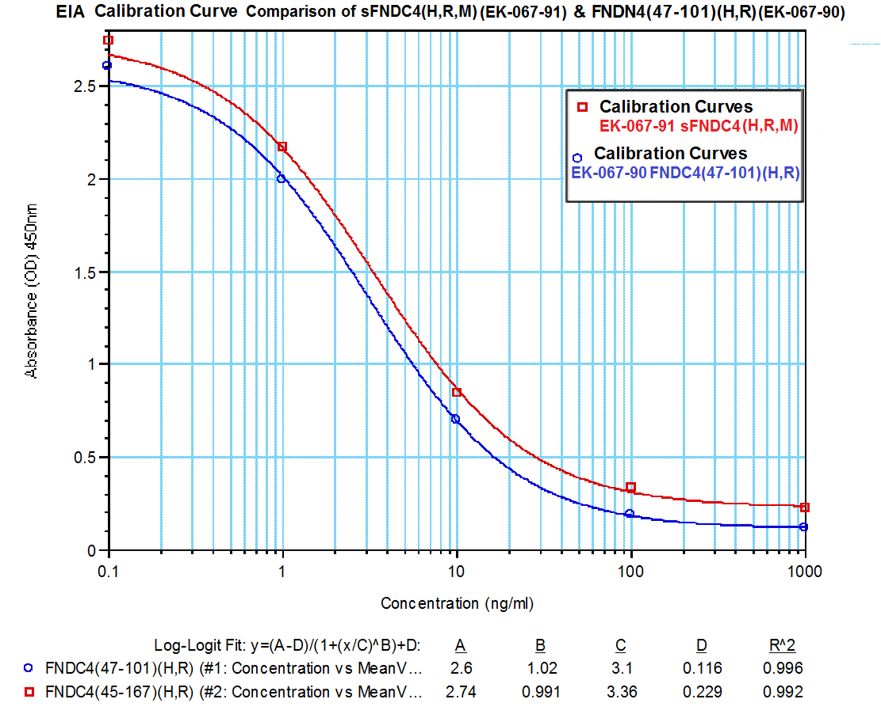
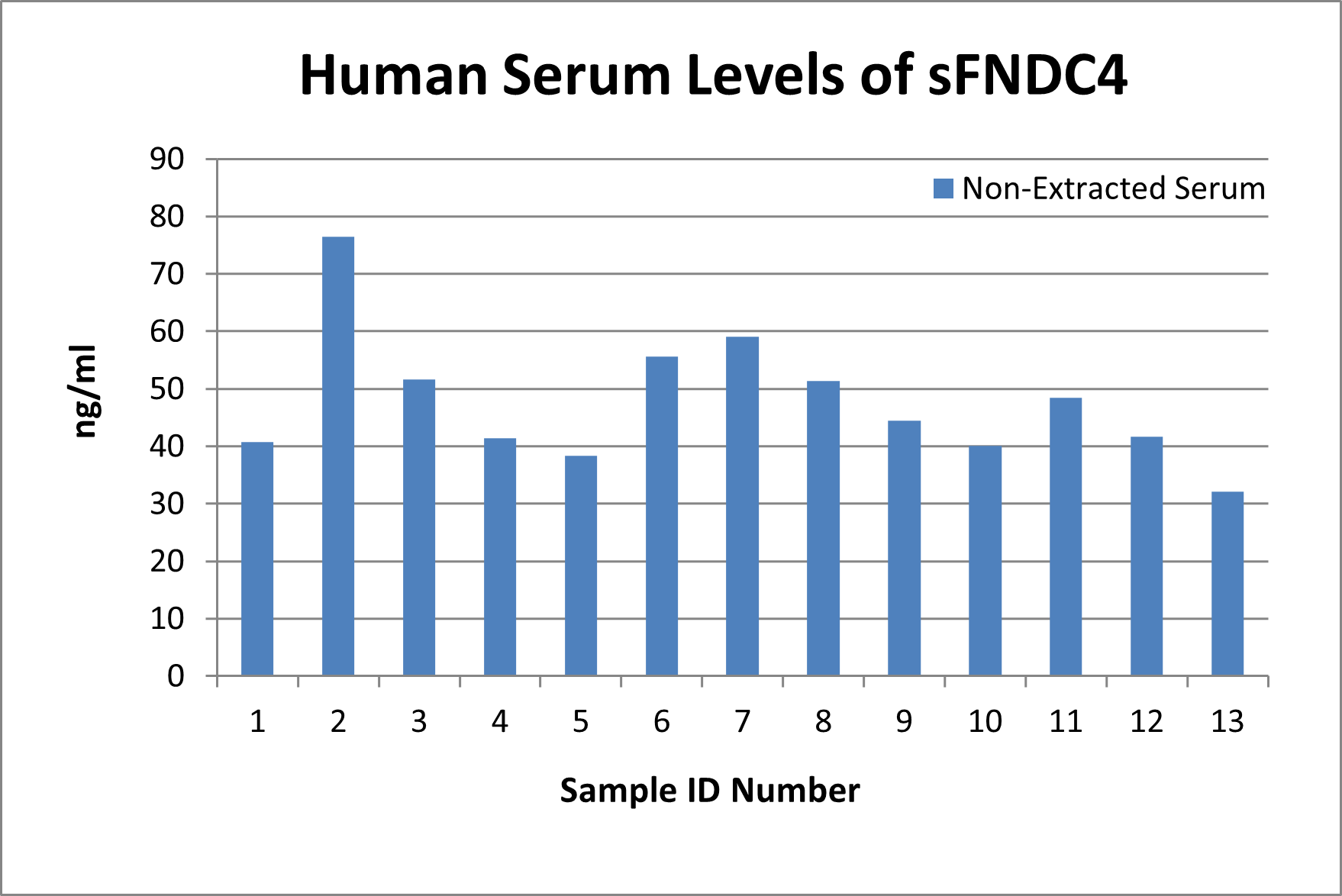
In-house measured levels of sFNDC4 using Catalog No. EK-067-91
Human Serum levels: 47.78 ± 3.06 ng/ml (n=13)
Abstract: The proper functional interaction between different tissues represents a key component in systemic metabolic control. Indeed, disruption of endocrine inter-tissue communication is a hallmark of severe metabolic dysfunction in obesity and diabetes. Here, we show that the FNDC4-GPR116, liver-white adipose tissue endocrine axis controls glucose homeostasis. We found that the liver primarily controlled the circulating levels of soluble FNDC4 (sFNDC4) and lowering of the hepatokine FNDC4 led to prediabetes in mice. Further, we identified the orphan adhesion GPCR GPR116 as a receptor of sFNDC4 in the white adipose tissue. Upon direct and high affinity binding of sFNDC4 to GPR116, sFNDC4 promoted insulin signaling and insulin-mediated glucose uptake in white adipocytes. Indeed, supplementation with FcsFNDC4 in prediabetic mice improved glucose tolerance and inflammatory markers in a white-adipocyte selective and GPR116-dependent manner. Of note, the sFNDC4-GPR116, liver-adipose tissue axis was dampened in (pre) diabetic human patients. Thus our findings will now allow for harnessing this endocrine circuit for alternative therapeutic strategies in obesity-related pre-diabetes
*The authors validate the quantified levels of sFNDC4 by using Phoenix’s FNDC4 (47-101) (Human) / FNDC4 (43-97) (Mouse) - EIA Kit (catalog #EK-067-90) and found zero cross-reactivity to irisin/FNDC5
Georgiadi A, Lopez-Salazar V, Merahbi RE-, et al. Orphan GPR116 mediates the insulin sensitizing effects of the hepatokine FNDC4 in adipose tissue. Nat Commun. 2021;12(1):2999.
Supplementary Information: https://static-content.springer.com/esm/art%3A10.1038%2Fs41467-021-22579-1/MediaObjects/41467_2021_22579_MOESM1_ESM.pdf
FNDC4 is a secreted factor sharing high homology with the exercise-associated myokine irisin (FNDC5). Here we report that Fndc4 is robustly upregulated in several mouse models of inflammation as well as in human inflammatory conditions. Specifically, FNDC4 levels are increased locally at inflamed sites of the intestine of inflammatory bowel disease patients. Interestingly, administration of recombinant FNDC4 in the mouse model of induced colitis markedly reduces disease severity compared with mice injected with a control protein. Conversely, mice lacking Fndc4 develop more severe colitis. Analysis of binding of FNDC4 to different immune cell types reveals strong and specific binding to macrophages and monocytes.FNDC4 treatment of bone marrow-derived macrophages in vitro results in reduced phagocytosis, increased cell survival and reduced proinflammatory chemokine expression. Hence, treatment with FNDC4 results in a state of dampened macrophage activity, while enhancing their survival. Thus, we have characterized FNDC4 as a factor with direct therapeutic potential in inflammatory bowel disease and possibly other inflammatory diseases.
Bosma M1, Gerling M2, Pasto J1 et al., Nat Commun. 2016 Apr 12;7:11314. doi: 10.1038/ncomms11314.
Many disorders are associated with altered serum protein concentrations, including malnutrition, cancer, and cardiovascular, kidney, and inflammatory diseases. Although these protein concentrations are highly heritable, relatively little is known about their underlying genetic determinants. Through transethnic meta-analysis of European-ancestry and Japanese genome-wide association studies, we identified six loci at genome-wide significance (p < 5 × 10(-8)) for serum albumin (HPN-SCN1B, GCKR-FNDC4, SERPINF2-WDR81, TNFRSF11A-ZCCHC2, FRMD5-WDR76, and RPS11-FCGRT, in up to 53,190 European-ancestry and 9,380 Japanese individuals) and three loci for total protein (TNFRS13B, 6q21.3, and ELL2, in up to 25,539 European-ancestry and 10,168 Japanese individuals). We observed little evidence of heterogeneity in allelic effects at these loci between groups of European and Japanese ancestry but obtained substantial improvements in the resolution of fine mapping of potential causal variants by leveraging transethnic differences in the distribution of linkage disequilibrium. We demonstrated a functional role for the most strongly associated serum albumin locus, HPN, for which Hpn knockout mice manifest low plasma albumin concentrations. Other loci associated with serum albumin harbor genes related to ribosome function, protein translation, and proteasomal degradation, whereas those associated with serum total protein include genes related to immune function. Our results highlight the advantages of transethnic meta-analysis for the discovery and fine mapping of complex trait loci and have provided initial insights into the underlying genetic architecture of serum protein concentrations and their association with human disease.
Rizkalla Salwa W, Edi Prifti, Aurélie Cotillard et al., et al., Am J Hum Genet. 2012 Oct 5;91(4):744-53. doi: 10.1016/j.ajhg.2012.08.021. Epub 2012 Sep 27.
Schizophrenia is one of the major psychiatric disorders. It is a disorder of complex inheritance, involving both heritable and environmental factors. DNA methylation is an inheritable epigenetic modification that stably alters gene expression. We reasoned that genetic modifications that are a result of environmental stimuli could also make a contribution. We have performed 26 high-resolution genome-wide methylation array analyses to determine the methylation status of 27,627 CpG islands and compared the data between patients and healthy controls. Methylation profiles of DNAs were analyzed in six pools: 220 schizophrenia patients; 220 age-matched healthy controls; 110 female schizophrenia patients; 110 age-matched healthy females; 110 male schizophrenia patients; 110 age-matched healthy males. We also investigated the methylation status of 20 individual patient DNA samples (eight females and 12 males. We found significant differences in the methylation profile between schizophrenia and control DNA pools. We found new candidate genes that principally participate in apoptosis, synaptic transmission and nervous system development (GABRA2, LIN7B, CASP3). Methylation profiles differed between the genders. In females, the most important genes participate in apoptosis and synaptic transmission (XIAP, GABRD, OXT, KRT7), whereas in the males, the implicated genes in the molecular pathology of the disease were DHX37, MAP2K2,FNDC4 and GIPC1. Data from the individual methylation analyses confirmed, the gender-specific pools results. Our data revealed major differences in methylation profiles between schizophrenia patients and controls and between male and female patients. The dysregulated activity of the candidate genes could play a role in schizophrenia pathogenesis.
Rukova B, Staneva R, Hadjidekova S, et al., Balkan J Med Genet. 2015 Apr 10;17(2):15-23. doi: 10.2478/bjmg-2014-0070. eCollection 2014.
The fibronectin type III (FNIII) repeat is one of three structural motifs originally identified in the fibronectin protein and has been well characterized in recent years. The consensus sequence has since been found in many different proteins including receptors and cell adhesion molecules. We report the cloning and expression analysis of Frcp1 and Frcp2, two members of a new FNIII repeat containing gene family. During embryonic development both genes are primarily expressed in the brain. In adult tissues, Frcp1 is strongly expressed in the liver and Frcp2 in the heart.Teufel A1, Malik N, Mukhopadhyay M et al., Gene. 2002 Sep 4;297(1-2):79-83.
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| EK-067-91 | sFNDC4 (Human, Rat, Mouse) - EIA Kit | 96 wells | $735 |
| EK-067-90 | FNDC4 (47-101) (Human) / FNDC4 (43-97) (Mouse) - EIA Kit | 96 wells | $624 |
| 067-91 | sFNDC4 (Human, Rat, Mouse) | 20 µg | $289 |
| 040-40 | ADGRF5 / GPR116 (1313-1346), Prepro (Human) | 100 µg | $284 |
| 067-93 | FNDC4 (102-158) (Human) / FNDC4 (98-154) (Mouse) | 100 µg | $407 |
| 067-92 | FNDC4 (103-158) (Human) / FNDC4 (99-154) (Mouse) | 100 µg | $407 |
| WBK-067-92 | FNDC4 (103-158) (Human) / FNDC4 (99-154) (Mouse) - Western Blot Kit | 1 kit | $777 |
| 067-94 | FNDC4 (190-234) / FRCP1 (190-234) (Human) | 100 µg | $382 |
| WBK-067-94 | FNDC4 (190-234) / FRCP1 (190-234) (Human) - Western Blot Kit | 1 kit | $777 |
| 067-90 | FNDC4 (47-101) (Human) / FNDC4 (43-97) (Mouse) | 100 µg | $382 |
Social Network Confirmation