Catalog # |
Size |
Price |
|
|---|---|---|---|
| MB-003-60 | 1 ml | $796 |



The present study was done to clarify whether intracerebroventricular (ICV) injection of cocaine- and amphetamine-regulated transcript (CART) affects feeding in chicks. ICV injection of CART significantly inhibited fasting-induced feeding of broiler chicks. In layer chicks, on the other hand, CART inhibited food intake in birds with ad libitum access to feed but only weakly affected intake of fasted birds. In addition, the ICV injection of CART attenuated neuropeptide Y (NPY)-induced feeding in both broiler and layer chicks. These results indicate that CART is one of the important regulators of feeding in chicks, but the suppressive effect on feeding is somewhat different between strains. Furthermore, the present study also demonstrates that CART interacts with NPY in the central nervous system to regulate feeding in chicks.
Tachibana T, Takagi T, Tomonaga S, et al. Neurosci Lett. 2003;337(3):131-4.
Cocaine- and amphetamine-regulated transcript (CART) peptide (CART-(55-102)) is involved in the suppression of food intake. We now report that CART-(55-102) is involved in anxiety in rodents. Intracerebroventricularly administered CART-(55-102) as well as intraperitoneal administration of N-methyl-beta-carboline-3-carboxamide (FG-7142), a selective GABA(A)/benzodiazepine receptor inverse agonist, reduced time spent in the open arms in the elevated plus-maze task in mice. CART-(55-102)-induced anxiogenic-like behavior in this task was attenuated by widely prescribed anxiolytics such as diazepam and buspirone. Likewise, CART-(55-102) and FG-7142 significantly reduced social interaction in mice. Both diazepam and buspirone significantly reversed CART-(55-102)-induced anxiogenic-like behavior in social interaction tests. By contrast, another biologically active CART peptide, CART-(62-102), was without effect in the elevated plus-maze task in mice. Moreover, intracerebroventricular administration of CART-(55-102) markedly increased the firing rate of locus coeruleus neurons in single unit recording in anesthetized rats. As CART-(55-102) produced anxiety-like effects in rodents, this peptide may possibly be involved in anxiety and stress-related behavior.
Chaki S, Kawashima N, Suzuki Y, Shimazaki T, Okuyama S. Eur J Pharmacol. 2003;464(1):49-54.
BACKGROUND: Cocaine- and amphetamine-regulated transcript (CART) is expressed within hypothalamic nuclei implicated in the regulation of feeding behaviour. It is up-regulated by leptin, and CART-derived peptides acutely inhibit food intake.
OBJECTIVE: The present study was designed to assess the long-term effects of central CART administration on food intake, body weight, plasma levels of glucose, insulin, leptin, free fatty acids and triglycerides, and on fuel utilisation in normal and high-fat-fed obese rats.
DESIGN: Normal and high-fat-fed obese rats were cannulated intracerebroventricularly (i.c.v.) and infused for 6 days with CART (55-102) or its vehicle. At day 4, animals were placed in an indirect calorimeter for a 24 h period during which the respiratory quotient and the energy expenditure were determined hourly.
RESULTS: In both normal and obese animals, the chronic i.c.v. infusion of CART (55-102) had marked, sustained inhibitory effects on food intake and body weight gain that were accompanied by decreases in plasma insulin and leptin levels. Using indirect calorimetry, it was observed that CART infusion promoted an increase in lipid oxidation in normal and in obese animals, although this increase reached statistical significance only in the obese group. The hypothalamic CART mRNA expression was found to be higher in obese rats (displaying hyperleptinaemia) than in normal animals.
CONCLUSION: The data together show that chronic i.c.v. CART infusion is effective in inhibiting food intake, favouring lipid oxidation and limiting fat storage, both in normal and high-fat-diet-induced obese rats. The CART pathway thus seems to be an important determinant of body weight homeostasis in normal animals as well as in a model of nutritionally induced obesity.
Rohner-jeanrenaud F, Craft LS, Bridwell J, et al. Int J Obes Relat Metab Disord. 2002;26(2):143-9.
Cocaine- and amphetamine-regulated transcript (CART) is expressed in the hypothalamus, and putative peptides encoded by CART potently inhibit feeding when administered centrally. CART is strongly down-regulated in the lateral hypothalamic area and the arcuate nucleus in animal models of obesity with disrupted leptin signaling. Here we have used in situ hybridization and immunohistochemistry to study CART expression in mice homozygous for the anorexia (anx) mutation which are characterized by a much reduced food intake and premature death. anx/anx mice had significantly decreased levels of CART mRNA label and peptide-immunoreactive cell bodies and fibers in the arcuate nucleus and a lower number of detectable CART-expressing cells in the dorsomedial hypothalamic nucleus/lateral hypothalamic area. Moreover, serum leptin levels were significantly lower in anx/anx mice compared to normal littermates, most likely due to the prominent depletion of body fat in these animals. The decrease in the anorexigenic agents leptin and CART, may reflect a compensatory down-regulation in response to the energy-deprived state of anx/anx mice. Alternatively, the reduced arcuate CART expression may be a consequence of a molecular defect in the arcuate nucleus of these animals.
Johansen JE, Broberger C, Lavebratt C, et al. Brain Res Mol Brain Res. 2000;84(1-2):97-105.
The mammalian hypothalamus strongly influences ingestive behaviour through several different signalling molecules and receptor systems. Here we show that CART (cocaine- and amphetamine-regulated transcript), a brain-located peptide, is a satiety factor and is closely associated with the actions of two important regulators of food intake, leptin and neuropeptide Y. Food-deprived animals show a pronounced decrease in expression of CART messenger RNA in the arcuate nucleus. In animal models of obesity with disrupted leptin signalling, CART mRNA is almost absent from the arcuate nucleus. Peripheral administration of leptin to obese mice stimulates CART mRNA expression. When injected intracerebroventricularly into rats, recombinant CART peptide inhibits both normal and starvation-induced feeding, and completely blocks the feeding response induced by neuropeptide Y. An antiserum against CART increases feeding in normal rats, indicating that CART may be an endogenous inhibitor of food intake in normal animals.Kristensen P, Judge ME, Thim L, et al. Hypothalamic CART is a new anorectic peptide regulated by leptin. Nature. 1998;393(6680):72-6.
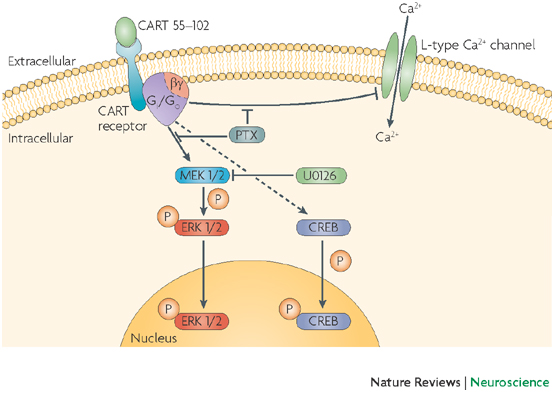
No References
| Catalog# | Product | Size | Price | Buy Now |
|---|
Social Network Confirmation